Obesity and Dementia
Obesity and Dementia: The Role of Adipocyte-Derived Exosomes in the Development of Alzheimer’s Disease
Sujay Ratna
Children’s National Medical Center
Thomas Jefferson High School for Science & Technology
This paper was originally included in the 2018 print publication of Teknos Science Journal.
Abstract
The overall objective for this study was to test the cross-sectional association between circulating adipocyte-derived exosomal microRNAs and the pathologic mechanisms underlying weight loss and Alzheimer’s disease (AD). miRNA could serve as potential biomarkers for AD, 15-20 years prior to the manifestation of symptoms due to exosomes in blood serum before they cross the blood-brain barrier. The researchers isolated adipocyte-derived exosomes from serum and CSF from patients with probable AD/dementia and controls from the Adult Changes in Thought (ACT) cohort. RNA was isolated from exosomes using mirVana miRNA Isolation Kit and then amplified using a SeraMir Exosome RNA Amplification Kit. Amplified microRNA was labeled using the FlashTag Biotin HSR RNA Labeling Kit and profiled on Affymetrix GeneChip miRNA 4.0 Arrays. In Ingenuity Pathways Analysis (IPA), mRNAs were highly predicted and experimentally-validated to be targeted by these microRNAs in both depots. These mRNAs were mapped to specific diseases and biological functions predicted to be affected. Serum and CSF microRNA expression strongly correlated with no microRNAs differing significantly. Comparison of AD versus control microRNA profiles identified eight microRNAs that were differentially expressed in both the serum and CSF with a highly conservative Fold Change ≥ |2|. The researchers found that adipocyte-derived exosomal microRNAs relevant to neurodegeneration are increased in serum from subjects with clinical dementia, decreased MRI hippocampal volumes, and increased in high AD neuropathological change as defined by CERAD scores and Braak stages. Importantly, the eight adipocyte-derived exosomal microRNAs expressed in dementia (vs. control) are known to activate pathways involved in neuronal degeneration and apoptosis.
Introduction
The overall objective for this study is to examine the correlation between circulating adipocyte-derived exosomal microRNAs in lean and obese patients with regards to their development of neurodegeneration. The researchers achieved this by using clinical data and biospecimens (e.g. serum, brain, and CSF) from participants (n=50 cases and 10 controls) in the University of Washington Alzheimer’s Disease Research Center (ADRC) Clinical Core as well as the Adult Changes in Thought (ACT) – Project 3. The study examined adipocyte-derived exosomal microRNAs in banked samples with analysis of the securely stored clinical data. The researchers tested their hypothesis in three aims:
To refine the circulating adipocyte-derived exosomal (ADE) microRNA signature for neurodegeneration by testing its association with poorer cognitive performance in AD/dementia (with analysis of cognitive subtypes using rich psychometric data in the ADRC Clinical Core).
Hypothesis 1: ADE microRNAs relevant to neurodegeneration will be increased in serum from subjects with clinical AD/dementia compared to those without.
To test the association between the circulating ADE microRNA signature for neurodegeneration and decreased MRI hippocampal volumes.
Hypothesis 2: ADE microRNAs relevant to neurodegeneration will be increased in serum from subjects with decreased MRI hippocampal volumes compared to those without.
To test the association between the circulating ADE microRNA signature for neurodegeneration and increased AD neuropathological change as well as changes of common comorbid conditions (e.g. Lewy body disease, vascular brain injury, hippocampal sclerosis, and TDP-43 pathology).
Hypothesis 3: ADE microRNAs relevant to neurodegeneration will be increased in the serum from subjects with intermediate or high AD neuropathological change as defined by CERAD scores and Braak stages compared to those with no or low AD neuropathological changes.
In sum, this project advances research on understanding mechanisms that may enable the primary prevention of cognitive decline and dementia, as recommended by the National Alzheimer's Plan [12] and the Alzheimer's Disease-Related Dementias Conference road maps [13].
Several studies have shown that exosome activity increases in patients with AD and highlighted the presence of exosomal microRNAs in blood. However, the relationship between circulating adipocyte-derived exosomal microRNAs and neuropathology has not been directly examined. Therefore, the hypothesis of this study is adipocyte derived exosomes will cross the blood brain barrier and be found in the CSF and will contain miRNAs targeting pathways involved in cognitive decline. This study aims to fill this gap by establishing relationships between circulating microRNAs and neuropathology in AD/dementia. This innovative research provides a novel mechanistic link between adipose tissue and neurological health.
Background
Obesity is a public health epidemic. Clinically obesity is defined as a BMI of greater than 30 kg/m2. In fact, obesity is a worldwide health problem, but specifically in the US, almost 37% of the population is obese (this number has increased from ~30% in 1999), and nearly 70% of the population is obese or overweight.
The association that the researchers were most interested in studying is that between adiposity and dementia/cognitive decline. Increased central adiposity is associated with an increased risk of cognitive decline in older persons with diabetes. Also, midlife obesity increases the risk of the development of dementia 30 years later obesity defined by waist circumference, due to the increase of visceral adiposity). Increased waist circumference associated with changes on fMRI that correlated with diminished working memory related BOLD response and slower task reaction time. In general, there have been some mixed findings regarding obesity and the development of dementia which likely has to do with the definition of BMI vs adiposity/central adiposity/visceral adiposity.
However, obesity’s role on the brain and cognitive function in adolescents is still unknown. Adolescents with metabolic syndrome demonstrate lower arithmetic, spelling, attention, and mental flexibility, as well as smaller hippocampal volumes and reductions in microstructural integrity of white matter tracts. Obese adolescents, even without insulin resistance have lower cognitive performance than lean adolescents. Thus, previous research shows that adipocyte-derived exosomes play a role in obesity and cognitive decline in adolescents, which may provide insight on biomarkers for Alzheimer’s, before symptoms occur.
Exosomes are cell derived vesicles 20-100 nanometers in size. They carry lipids, sugars, nucleic acids, and proteins and are extremely important in cell communication. Adipocyte-derived exosomes contain potential signal mediators (miRNA) capable of instigating end organ tissue dysfunction. Exosomes are a means of intracellular signaling, and carry microRNAs (miRNAs) that inhibit messenger RNA (mRNA) translation or increase mRNA degradation. The accumulation of visceral adipose tissue (VAT) in specific is linked to cardiometabolic diseases, including diabetes, dyslipidemia, hypertension and heart disease, as shown in Figure 1. VAT can release exosomes into circulation which go on to affect gene expression in end organs. Liver associated with development of NAFLD, in skeletal muscle.
Alzheimer’s disease (AD) is the most common neurodegenerative dementia [1], with symptoms of gradual memory loss and deterioration of behavior, language, and orientation [2]. Biologically, AD brains have deposition of both amyloid-beta plaques and total/phosphorylated tau tangles [3]. Currently, researchers use biomarkers such as cerebrospinal fluid (CSF) and results from MRI and PET scans to pinpoint the progression of AD [4, 5].
However, a new potential candidate for an AD biomarker has been identified: microRNAs (miRNAs) [6]. miRNAs are endogenous noncoding ribonucleic acids, that modulate gene expression at the post-transcriptional level [7]. MicroRNAs inhibit the translation of mRNA by promoting their degradation [8]. Each microRNA targets hundreds of genes while each individual gene is targeted by multiple microRNAs. They are transcribed in the nucleus and afterwards they are exported to the cytoplasm, via exosomes [9]. In fact, scientists have noted similar miRNA sequences in blood serum and plasma and CSF in AD patients, which could suggest the role of exosomes in transporting miRNAs in AD [10, 13]. Some of those microRNAs are involved in different cellular functions such as neurogenesis, cell proliferation, immune response, or microglial mediated neuronal injury [15]. However, the lack of reproducible results across studies still question the role of microRNAs as potential biomarkers for AD [9].
Dementia pathogenesis is clearly multifactorial, related to chronic conditions common in middle age including hypertension/cardiovascular disease, type 2 diabetes mellitus (T2DM), and cerebral macrovascular and microvascular disease. In fact, central obesity in middle age is associated with a three-fold increased risk of dementia 30 years later [8]. This raises the possibility that an obesity-related factor is present in middle age that either directly or indirectly disrupts central nervous system homeostasis.
Neurodegeneration in AD is thought to begin years before clinical dementia, which is divided into three phases: 1) preclinical, 2) mild cognitive impairment (MCI), and 3) clinical dementia [14, 15, 16]. The mechanism by which adiposity/obesity raises risk for AD/dementia is largely unknown. Prior research has focused on adipokines. A new possibility is adipocyte-derived exosomes. Animal studies show that exosomes cross the blood brain barrier [21, 22]. The researchers have developed a method to isolate adipocyte-derived exosomes and has demonstrated a role for these exosomes in the development of systemic disease [10, 11].
Structural findings associated with AD include volume loss of the hippocampus and entorhinal cortex [10]. Furthermore, decrease in structural hippocampal volume and hippocampal surface changes are associated with other biomarkers of AD/dementia [12]. Structural changes can even be demonstrated in preclinical disease [12, 13]. In patients with MCI, hippocampal atrophy is associated with conversion to AD [13].
The hallmark lesions of AD include amyloid beta deposition in senile plaques, neuritic plaques and neurofibrillary tangles [14]. Each of these markers has individual scoring systems. However, the ABC score incorporates histopathologic assessments of amyloid beta deposits, staging of neurofibrillary tangles, and scoring of neuritic plaques [29]. Montine et al. suggested a practical application for the ABC score to assign levels of AD neuropathologic change [15].
Materials and Methods
This study utilized measurements and data from the “Adult Changes in Thought” (ACT) Cohort. The Prospective Cohort Study Begun in 1994, and gathered members of a community-based health maintenance organization in Seattle. 5422 patients who were 65 years or older, non-nursing home residents were eligible and 2581 were enrolled into the longitudinal study. From this Cohort, screening for dementia was performed initially and then every 2 years. This included samples of Serum and CSF from these individuals. The data used in this study came from 5 paired CSF and serum samples from patients with Alzheimer’s disease and 3 paired samples from patients without Alzheimer’s disease. This made up the case patient and control subject group for the current study.
As shown in Figure 5, the controls in this study did not have clinical dementia. Though there is a significant difference in the dementia group versus the controls, these controls are not really biologically controls. They just do not have clinical dementia, so they mix together in the PCA plots. The experimental cases and controls of the study are defined as:
Cases:
1) Age 65-91 years;
2) Probable AD dementia: meets core clinical criteria;
3) Three levels of evidence of AD pathophysiological process (intermediate/high biomarkers, positive Aβ (PET or CSF) and/or positive neuronal injury (CSF tau, FDG-PET, structural MRI)).
Controls:
1) Age 65-91 years;
2) no history of cognitive decline from previous level of normal function;
3) MMSE score>28; 4) CDR (clinical dementia rating) score=0.
University of Washington ADRC collected blood and CSF from the selected patients in the ACT cohort and shipped them to the researchers, after de-identifying patient data. Whenever the patients would visit their neurologist for a checkup, they were scheduled for lab test. The researchers included data from interviews with physician, medical records, and laboratory tests. CSF was acquired from ventriculostomy at the time of autopsy. The CSF sample was spun down at 3000xg for 15 minutes to rid it of cell debris and unwanted proteins. During the same visit, patients’ blood was taken and immediately spun down using a centrifuge at 1000xg for 15 minutes. The resulting supernatant was designated as serum and was immediately aliquoted and stored at -80 degrees Celsius in a freezer.
After CSF samples were obtained from an affiliated university, the researchers proceeded with the exosome extraction step, using protocols outlined by Systems Biosciences. A summary of the methods is outlined in Figure 3. In essence, the researchers isolated exosomes from the serum using ExoQuick and added FABP4 to the solution of adipocyte-derived exosomes. The researchers discovered that adipocyte derived exosomes contain FABP4 on phospholipid membrane was a differentiated adipocyte marker. Using a magnetic bead containing antibody for FABP4, the researchers captured the isolated exosomes of adipocyte origin from circulation/blood/body fluids magnetically. This process is depicted in Figure 3, which includes the quantification step of the exosome isolation. Stored plasma/serum specimens were processed for blood collection and adipocyte-derived exosome isolation. Total exosomes were isolated using ExoQuick exosome precipitation solution. Adipocyte-derived exosomes were positively-selected by targeting fatty acid binding protein 4 (FABP4) [11].
Quantification of adipocyte-derived exosomes was done through magnetic isolation with beads and FABP4. The resulting bondage was counted through a flow cytometry machine with first class lasers. Figure 2 portrays the frequency that the laser hit the magnetic bead marker for the antigen that was bound to each exosome. Once quantified, the researchers stored the flow buffer with the exosomes in it, in a freezer at -80 degrees Celsius. This process was repeated for the CSF samples.
The researchers then ran a microRNA array from Qiagen on the CSF and serum adipocyte-derived exosomes. Over 7000 individual markers tested the miRNA concentration by fluorescent markers that were proportional to the amount of gene expression. This data was run through Partek to look for differences in Alzheimer’s disease patients versus the controls. Including p-values, the researchers calculated fold change, a measure of how many times the difference between the case and controls was. A significant fold change (FC) value was deemed as greater than 2.0 in either direction, positive or negative. Finally, the researchers utilized the Ingenuity Pathway Analysis (IPA) software to predict mRNA targets and pathways. Causal networks, heat maps, and principal component analysis was conducted on the IPA data.
The researchers compared dementia cases and controls among the 1,033 subjects in the Strong Heart Stroke Study for circulating adipocyte-derived exosomal microRNA signatures for T2DM at original Strong Heart Study enrollment. They then compared dementia cases and controls among the 1,033 subjects in the Strong Heart Stroke Study for circulating adipocyte-derived exosomal microRNA signatures for atherosclerotic macrovascular disease at original Strong Heart Study enrollment. By integrating circulating adipocyte-derived exosomal microRNA signatures and biochemical measures of T2DM/atherosclerotic macrovascular disease during middle age with dementia phenotyping data, the researchers determined dementia endotypes with the use of an innovative systems-based approach.
Results and Discussion
Differential Expression Analyses, with covariates, were performed using function for linear model fitting in the limma R package [16]. The significance of differences in microRNA expression was tested using an empirical Bayes moderated t-test, and the results were compared with the less powerful Wilcoxon rank sum test without covariates. Correction for multiple testing was addressed by controlling the false discovery rate (FDR) using the Benjamini and Hochberg (BH) method. A probe was considered significantly differentially expressed if the BH-corrected P was < 0.05. All reported differentially expressed probes were significant at P < 0.05 using the rank sum test. Using R Statistical Software, the researchers generated a heatmap of mean-centered normalized expression values and an unsupervised hierarchical clustering of study subjects based on Euclidean distances of expression profiles was performed to assess grouping. In addition to identifying transcripts that are differentially expressed between the cases and controls, the researchers explored development of an integrated sparse regression model to discriminate the two groups using a LASSO logistic regression approach and employed cross-validation both to calibrate the sparsity of the regressors and the error rate of the model. Lists of microRNAs were analyzed using Ingenuity Pathway Analysis software to identify significantly altered pathways. The annotation database was used to annotate the biological function, processes and pathways associated for each gene under analysis. Ingenuity Pathway Analysis uses right tailed Fisher exact test to derive P-values for identifying significantly over-represented pathways.
The researchers started with a sample size of 50 cases and 10 controls and used this sample size for all power calculations. Power calculations were performed using SPSS version 22 and the R package power. Based on preliminary data, gene expression has a coefficient of variation <2 and accounting for potential sample dropout of 10%, the sample size has >80% power to detect differential expression that is >1.6-fold between outcome groups.
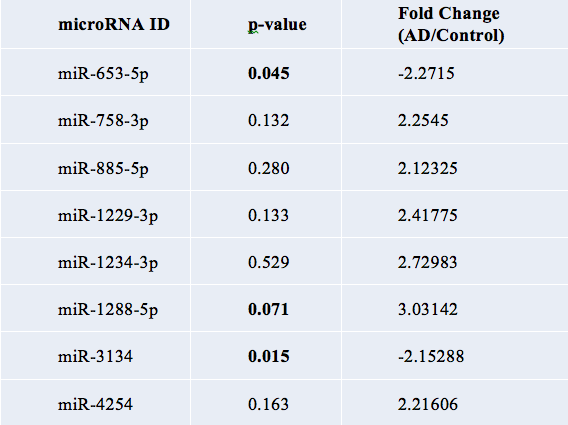
In hypothesis exploring tests, small sample size means p-value is irrelevant, so the researchers used fold change to determine significant data. 84 miRNAs with absolute value FC 2 or greater difference between AD and Control were found. This yielded 7701 analysis ready-molecules when the researchers applied mRNA target filter using only those highly predicted, experimentally observed miRNAs. If the difference between the cases and controls is bigger, then it is more likely to be real. Of the 84 miRNAs that were significant, 28 miRNAs met that criteria of fold change 2.0 or greater, and some even had significant p-values, when overlapped with the 22 significant in CSF exosomes and 56 from serum.
Figure 4 shows the checked residual distributions and Q-Q plots for appropriateness of linear analysis. This shows that FABP4+ exosomes (ADE) can be isolated from CSF and Serum and show nearly identical expression profiles. Of the 2700 probes tested, only 118 show different expression patterns between the biofluids (only 118 fell outside of the confidence interval). Therefore, in the future, serum can be used to study the adipocyte-derived exosomes in CSF. This has never been shown before in previous literature, and the researchers linear plot shows the confidence they have in exosomes crossing the blood-brain barrier while transporting AD relevant miRNA. The results also suggest that Alzheimer’s may be treated before plaques and biomarkers accumulate in the brain, as they travel in the bloodstream before then (15-20 years in advance).
Due to the imprecise nature of shipping the serum and CSF samples from the affiliated university to the research institution, the samples arrived on different days. Unfortunately, this meant that processing would occur on two different days as well. Because of the batch effect, the researchers conducted a PCA plot to remove the batch effect and normalize the data so human error or aged reagents would not affect exosome quantification. Figure 6 depicts the overlap between the two batches. It can be seen that there is some overlap in the different days, but overall, PCA shows there is a difference in processing on different days. However, the PCA correlation coefficient is only 61% so there is no significant difference.
Figure 7 exhibits a heat map of the hierarchical clustering of the Clustering of CSF. miRNAs with an absolute value of FC 2 or greater in CSF are clustered on rows and columns. In the ‘y-axis’, the second column has black boxes which stand for Alzheimer’s Disease patients and pink for the controls. As expected, the black and the pink controls and they mix in with each other, showing the miRNAs in CSF are upregulated.
The top of miRNAs cluster into bigger groups: the red along that bottom right quadrant means that they're all they're upregulated in those subjects, green means they are downregulated so there's clearly a pattern of differences. The principal components analysis (PCA) on CSF in Figure 8 shows reversible components for each patient part. The cases are the ovals, controls are the boxes, ones that say L.B. are Lewy body, and the main result is that they do not separate out.
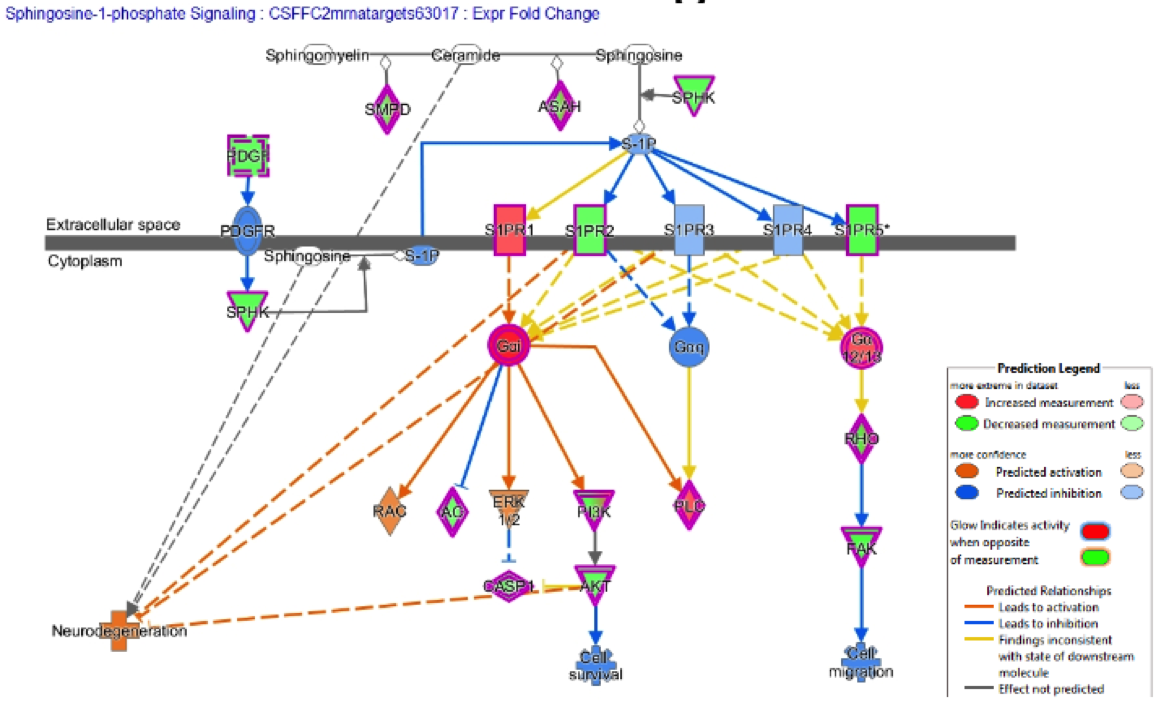
One explanation of the results is the Sphingosine 1 Phosphate Receptor Signaling pathway that is predicted to be upregulated, as shown in Figure 9. The orange connections mean the prediction of upregulation of apoptosis and is in line with the migration of axons during neurodevelopment. In addition, RhoGDI related to oxidative stress and NADPH oxidation function. So, three biologically relevant pathways that actually have literature and to mention also those diseases that are predicted to be altered in Alzheimer’s disease.
Exosomes are actually going across the board brain barrier and they are affecting CNS functions whether it is older people related to dementia or younger people related to cognitive function. mRNAs that are downregulated by the miRNAs during translation so less protein produced from the mRNA and IPA predicted certain pathways to be increased or decreased and five pathways increased, all related to cell death. In fact, MRI data on brain mass/size supports these pathways’ prediction. Neuronal connective and vascular tissues meaning there are direct and indirect effects of these exosomes in the CNS.
After conducting a literature review on the 8 miRNAs as potential biomarkers of AD, one microRNA stood out. In a study of miRNA expression in 7 patients with Alzheimer’s disease, miR-137 was also down-regulated in the frontal cortex. Suppression of SPTLC1 by miR-137 and miR-181c reduced Aβ expression levels in a target specific manner while over-expression of SPTLC1 and inhibition of miR-137 and miR-181c increased Aβ expression levels. In fact, the researchers concluded miR-137 was down-regulated in the blood serum of probable AD patients.
The first specific aim is to test circulating adipocyte-derived exosomal microRNA signatures (for T2DM) at enrollment for prospective association with dementia 15-25 years later. At enrollment, high expression of circulating adipocyte-derived exosomal microRNAs that are predicted to impair insulin signaling is associated with higher risk of developing dementia. The researchers also want to test circulating adipocyte-derived exosomal microRNA signatures (for atherosclerotic macrovascular disease) at enrollment for prospective association with dementia 15-25 years later. In addition, those that induce atherosclerotic macrovascular disease are associated with higher risk of developing dementia. In the future, the researchers can derive dementia endotypes in the Strong Heart Stroke Study of miRNA profiles with respect to lean and obese patients.
Conclusion
The results of this study show potential microRNA biomarkers for Alzheimer’s disease that are shown in Figure 10. The miRNAs are derived from adipose fat tissue which has a direct correlation with BMI and is related to obesity. The researchers found that apoptosis of neurons, cell death, dysgenesis, growth failure, hypoplasia of organs, movement disorders, morbidity, and neonatal/organismal/perinatal death are all disease and functions that are up regulated in the Alzheimer’s patients in this study.
One major limitation of this study was sample size. A larger population will be available to study through the Strong Heart Study. This gives the researchers the opportunity to analyze the difference between obesity and Alzheimer’s disease in the general population. More clinical information and imaging would be beneficial in confirming and extrapolating the results of this current study. In addition, the controls must be more than clinically clean. Rather than having only two groups, Alzheimer’s disease can be split into forms of dementia, with a group for Lewy Body Disease, neuritic plaques, Parkinson’s disease, etc. Since the control patients have some histologic markers of AD pathology, it will be a challenge to obtain additional controls, though may be difficult to obtain large number of controls without any neuritic plaques.
In future studies, the researchers desire to obtain CSF samples over a long period of time, not just at the time of death. For the most efficient accession to CSF, a spinal tap or lumbar puncture must be used. This method would allow the researchers to investigate the effects of exosome trafficking in obesity and Alzheimer’s disease over a span of 15 to 20 years. The researchers would like to calculate Body Mass Index and weight trend information for the study subjects. BMI can be calculated from height and weight measurements which are provided for six of the subjects. However, only four of these subjects have measurements recording for multiple visits, so only four graphs indicating change in BMI over time can be made. One final confirmation the researchers would like to explore is functional testing, in which they can measure actual mRNA in brain tissue. In vitro studies have explored incubating neurons with ADE to demonstrate that they enter neurons, and gene expression changes can give insight into the miRNA profile.
References
[1] Prince, M., et al. World Alzheimer Report 2015. The global impact of dementia: an analysis of prevalence, incidence, cost and trends. Alzheimer's Disease International. 2015; Available from: https://www.alz.co.uk/research/WorldAlzheimerReport2015.pdf.
[2] Aisen, P.S., et al., On the path to 2025: understanding the Alzheimer's disease continuum. Alzheimers Res Ther, 2017. 9(1): p. 60.
[3] Kivipelto, M., et al., Risk score for the prediction of dementia risk in 20 years among middle aged people: a longitudinal, population-based study. The Lancet Neurology, 2006. 5(9): p. 735-741.
[4] Ott, A., et al., Diabetes mellitus and the risk of dementia: The Rotterdam Study. Neurology, 1999. 53(9): p. 1937-42.
[5] Gorelick, P.B., et al., Vascular contributions to cognitive impairment and dementia: a statement for healthcare professionals from the american heart association/american stroke association. Stroke, 2011. 42(9): p. 2672-713.
[6] Mayeda, E.R., et al., Racial/ethnic differences in dementia risk among older type 2 diabetic patients: The diabetes and aging study. Diabetes care, 2014. 37(4): p. 1009-1015.
[7] Whitmer, R., et al., Central obesity and increased risk of dementia more than three decades later. Neurology, 2008. 71(14): p. 1057-1064.
[8] Sergi, G., et al., Weight loss and Alzheimer's disease: temporal and aetiologic connections. Proc Nutr Soc, 2013. 72(1): p. 160-5.
[9] Cova, I., et al., Weight Loss Predicts Progression of Mild Cognitive Impairment to Alzheimer's Disease. PLoS One, 2016. 11(3): p. e0151710.
[10] Hubal, M.J., et al., Circulating adipocyte-derived exosomal MicroRNAs associated with decreased insulin resistance after gastric bypass. Obesity (Silver Spring), 2017. 25(1): p. 102-110.
[11] Ferrante, S.C., et al., Adipocyte-derived exosomal miRNAs: a novel mechanism for obesity-related disease. Pediatr Res, 2015. 77(3): p. 447-54.
[12] Alzheimer's Association National Plan Milestone, W., et al., 2014 Report on the Milestones for the US National Plan to Address Alzheimer's Disease. Alzheimers Dement, 2014. 10(5 Suppl): p. S430-52.
[13] Montine, T.J., et al., Recommendations of the Alzheimer's disease-related dementias conference. Neurology, 2014. 83(9): p. 851-60.
[14] Sperling, R.A., et al., Toward defining the preclinical stages of Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement, 2011. 7(3): p. 280-92.
[15] Albert, M.S., et al., The diagnosis of mild cognitive impairment due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimer's & dementia, 2011. 7(3): p. 270-279.
[16] McKhann, G.M., et al., The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimer's & dementia, 2011. 7(3): p. 263-269.